Center for Pharmaceutical Value (PValue)
Pharmaceutical Value (pValue) - headquartered within the University of Colorado's Anschutz Medical Campus - aims to apply and test novel methods for value assessment that encourages stakeholder engagement and promotes value-based decision making. pValue is a recipient of the PhRMA Foundation's Value Assessment Initiative Centers of Excellence Award.
Mission Statement
Pharmaceutical Value (pValue)’s mission is to apply and test novel methods to improve pharmaceutical coverage and reimbursement decision-making.
Team

R. Brett McQueen PhD
Associate Professor
- Department of Clinical Pharmacy
Email Address:robert.mcqueen@cuanschutz.edu
Primary Phone:303-724-0251
Mailing Address:
- CU Anschutz
Pharmacy and Pharmaceutical Sciences Building
12850 East Montview Boulevard
First Floor Room 1205
Aurora, CO 80045
Dr. McQueen is an Assistant Professor at the University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences (CU-SSPPS). He trained at CU-SSPPS in pharmacoeconomics, and after a position at the Observational and Pragmatic Research Institute in Cambridge, UK, he re-joined CU-SSPPS as faculty. His funding lines include comparative effectiveness research, cost-effectiveness applications and methods development, value-based outcomes contracting, and patient preferences research. He is active in health economics-related societies such as the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) through contributions to ISPOR short courses, workshops, issue panels, and research presentations. He is the course director for a PhD course titled, “Pharmaceutical Economics.” Dr. McQueen serves on multiple dissertation committees in the Pharmaceutical Outcomes Research PhD program. Finally, he is currently the director of the Center for Pharmaceutical Value (pValue), exploring multi-criteria decision analysis (MCDA) for health care decision making.
Professional Service:
Brett served in a PhRMA Foundation Postdoctoral Fellowship in Health Outcomes at SSPPS and most recently was a Health Economist and Researcher with the Research in Real Life (RiRL)/Observational and Pragmatic Research Institute (OPRI) located in Cambridge, United Kingdom.
Education, Licensure & Certifications
- BA - Economics, University of Colorado, Boulder
- MA - Economics, University of Colorado, Denver
- PhD - Pharmacoeconomics, University of Colorado, Denver
Partners with pValue


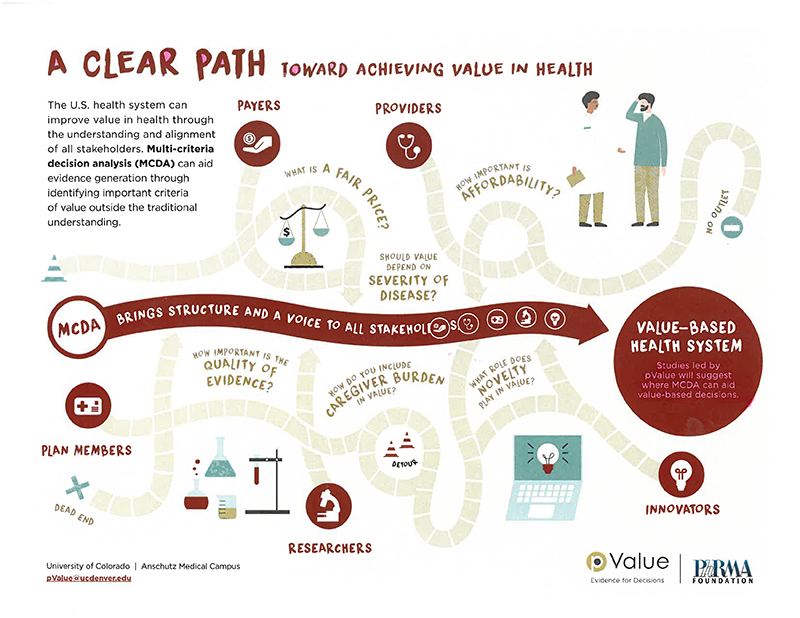
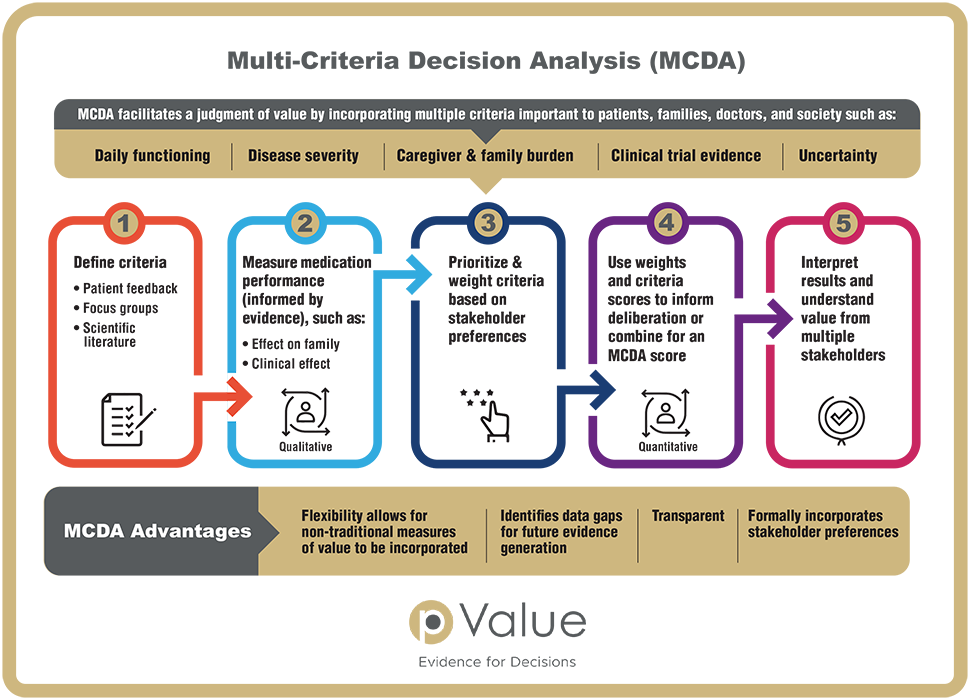
Traditional measures of value used in cost-effectiveness analyses are not, by definition, fully comprehensive or sufficiently flexible to allow for the inclusion of all the criteria that patients, payers, clinicians, or other health care stakeholders care about. Multi-criteria decision analysis (MCDA) offers a scientifically rigorous decision-making tool capable of including multiple criteria that are important to stakeholders. MCDA has been applied in a variety of sectors, such as investment banking and environmental management, but applications in US healthcare decision making have been limited.
MCDA can improve decision making in health care by engaging key stakeholders and by capturing and weighing criteria not found in traditional measures of value, for example novelty, severity of disease, quality of evidence, and family burden. By encouraging a comprehensive understanding of value, MCDA offers an opportunity to systematically weigh non-traditional aspects of value that fall outside traditional measures of value.
Publications

Complementing Coverage and Reimbursement Decisions With Multi-Criteria Decision Analysis
Featuring Dr. Brett McQueen and Dr. Jon Campbell
Press

Industry Voices—Using PCORI to advance patient-centered values
Featuring Dr. Brett McQueen

CU Anschutz investigators receive PhRMA Foundation Award
Featuring Dr. Brett McQueen and Dr. Jon Campbell